| NDC | 69543-223-30 |
| Set ID | 2521782a-87a0-41d5-9d2f-41c5015b778c |
| Category | DIETARY SUPPLEMENT |
| Packager | Virtus Pharmaceuticals |
| Generic Name | |
| Product Class | |
| Product Number | |
| Application Number |
-
HEALTH CLAIM
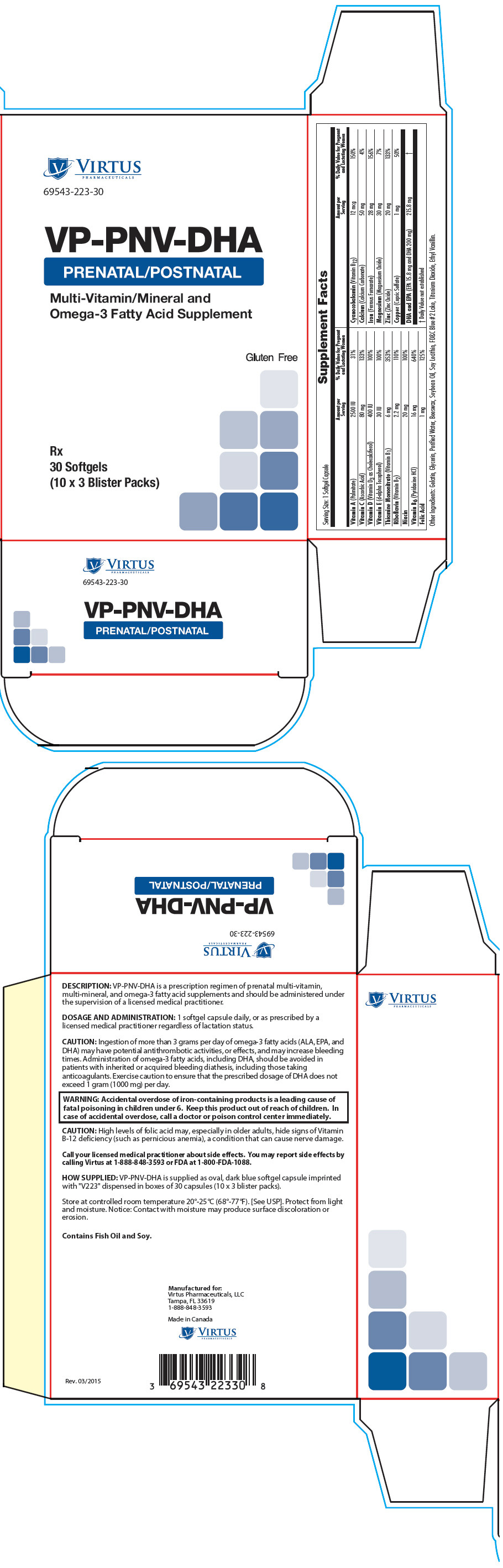
Supplement Facts Serving Size: 1 Softgel Capsule Amount per Serving % Daily Value for Pregnant and Lactating Women - *
- Daily Value not established
Vitamin A (Palmitate) 2500 IU 31% Vitamin C (Ascorbic Acid) 80 mg 133% Vitamin D (Vitamin D3 as Cholecalciferol) 400 IU 100% Vitamin E (d-alpha Tocopherol) 30 IU 100% Thiamine Mononitrate (Vitamin B1) 6 mg 353% Riboflavin (Vitamin B2) 2.2 mg 110% Niacin 20 mg 100% Vitamin B6 (Pyridoxine HCl) 16 mg 640% Folic Acid 1 mg 125% Cyanocobalamin (Vitamin B12) 12 mcg 150% Calcium (Calcium Carbonate) 50 mg 4% Iron (Ferrous Fumarate) 28 mg 156% Magnesium (Magnesium Oxide) 30 mg 7% Zinc (Zinc Oxide) 20 mg 133% Copper (Cupric Sulfate) 1 mg 50% DHA and EPA (EPA 15.8 mg and DHA 200 mg) 215.8 mg * Other Ingredients: Gelatin, Glycerin, Purified Water, Beeswax, Soybean Oil, Soy Lecithin, FD&C Blue #2 Lake, Titanium Dioxide, Ethyl Vanillin.
- DESCRIPTION
- DOSAGE AND ADMINISTRATION
-
CAUTION
Ingestion of more than 3 grams per day of omega-3 fatty acids (ALA, EPA, and DHA) may have potential antithrombotic activities, or effects, and may increase bleeding times. Administration of omega-3 fatty acids, including DHA, should be avoided in patients with inherited or acquired bleeding diathesis, including those taking anticoagulants. Exercise caution to ensure that the prescribed dosage of DHA does not exceed 1 gram (1000 mg) per day.
- WARNINGS
- CAUTION
- HEALTH CLAIM
- HOW SUPPLIED
- SAFE HANDLING WARNING
- HEALTH CLAIM
- HEALTH CLAIM
- PRINCIPAL DISPLAY PANEL - 30 Softgel Blister Pack Carton
-
INGREDIENTS AND APPEARANCE
VP-PNV-DHA
vitamin a palmitate, ascorbic acid, cholecalciferol, .alpha.-tocopherol, d-, thiamine, riboflavin, niacin, pyridoxine hydrochloride, folic acid, cyanocobalamin, calcium carbonate, ferrous fumarate, magnesium oxide, zinc oxide, cupric sulfate, doconexent, and icosapent capsule, liquid filledProduct Information Product Type DIETARY SUPPLEMENT Item Code (Source) NHRIC:69543-223 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength Vitamin A Palmitate (UNII: 1D1K0N0VVC) (Vitamin A - UNII:81G40H8B0T, Retinol - UNII:G2SH0XKK91) Vitamin A 2500 [iU] Ascorbic Acid (UNII: PQ6CK8PD0R) (Ascorbic Acid - UNII:PQ6CK8PD0R) Ascorbic Acid 80 mg Cholecalciferol (UNII: 1C6V77QF41) (Cholecalciferol - UNII:1C6V77QF41) Cholecalciferol 400 [iU] .Alpha.-Tocopherol, D- (UNII: N9PR3490H9) (.Alpha.-Tocopherol, D- - UNII:N9PR3490H9) .Alpha.-Tocopherol, D- 30 [iU] Thiamine (UNII: X66NSO3N35) (Thiamine Ion - UNII:4ABT0J945J) Thiamine 6 mg Riboflavin (UNII: TLM2976OFR) (Riboflavin - UNII:TLM2976OFR) Riboflavin 2.2 mg Niacin (UNII: 2679MF687A) (Niacin - UNII:2679MF687A) Niacin 20 mg Pyridoxine Hydrochloride (UNII: 68Y4CF58BV) (Pyridoxine - UNII:KV2JZ1BI6Z) Pyridoxine 16 mg Folic Acid (UNII: 935E97BOY8) (Folic Acid - UNII:935E97BOY8) Folic Acid 1 mg Cyanocobalamin (UNII: P6YC3EG204) (Cyanocobalamin - UNII:P6YC3EG204) Cyanocobalamin 12 ug Calcium Carbonate (UNII: H0G9379FGK) (CALCIUM CATION - UNII:2M83C4R6ZB) CALCIUM CATION 50 mg Ferrous Fumarate (UNII: R5L488RY0Q) (Ferrous Cation - UNII:GW89581OWR) Ferrous Cation 28 mg Magnesium Oxide (UNII: 3A3U0GI71G) (Magnesium Cation - UNII:T6V3LHY838) Magnesium Cation 30 mg Zinc Oxide (UNII: SOI2LOH54Z) (ZINC CATION - UNII:13S1S8SF37) ZINC CATION 20 mg Cupric Sulfate (UNII: LRX7AJ16DT) (Cupric Cation - UNII:8CBV67279L) Cupric Cation 1 mg Doconexent (UNII: ZAD9OKH9JC) (Doconexent - UNII:ZAD9OKH9JC) Doconexent 200 mg Icosapent (UNII: AAN7QOV9EA) (Icosapent - UNII:AAN7QOV9EA) Icosapent 15.8 mg Inactive Ingredients Ingredient Name Strength Gelatin (UNII: 2G86QN327L) Glycerin (UNII: PDC6A3C0OX) Water (UNII: 059QF0KO0R) Yellow Wax (UNII: 2ZA36H0S2V) Soybean Oil (UNII: 241ATL177A) Lecithin, Soybean (UNII: 1DI56QDM62) FD&C Blue No. 2 (UNII: L06K8R7DQK) Aluminum Oxide (UNII: LMI26O6933) Titanium Dioxide (UNII: 15FIX9V2JP) Ethyl Vanillin (UNII: YC9ST449YJ) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NHRIC:69543-223-30 3 in 1 CARTON 1 10 in 1 BLISTER PACK Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date DIETARY SUPPLEMENT 08/25/2015 Supplement Facts Serving Size : Serving per Container : Amount Per Serving % Daily Value color scoring 1 shape size (solid drugs) 23 mm imprint Labeler - Virtus Pharmaceuticals (079659493)