| NDC | 50814-010-01, 50814-011-01, 50814-012-01, 50814-013-01, 50814-014-01, 50814-015-01, 50814-020-01 |
| Set ID | 94dd796a-d589-4744-b11a-acc8ef4b346f |
| Category | HUMAN OTC DRUG LABEL |
| Packager | GFA Production (Xiamen) Co., Ltd. |
| Generic Name | |
| Product Class | Amide Local Anesthetic, Aminoglycoside Antibacterial, Antiarrhythmic |
| Product Number | |
| Application Number | PART333B |
- Drug Facts
- Active ingredient
- Use
-
Warnings
For external use only.
When using this product
• to avoid contamination, do not touch tip of container to any surface • do not reuse • once opened, discard • obtain immediate medical treatment for all open wounds in or near the eyes
- Directions
- Other information
- Inactive ingredients
- DRUG FACTS
- Active Ingredient:
- Use:
- Warnings:
- Directions:
- Inactive ingredient:
- DRUG FACTS
- Active Ingredient:
- Use:
- Warnings:
- Directions:
- Other information:
- Inactive ingredient:
- Drug Facts
- Active ingredients (in each gram)
- Use
- Warnings
- Directions
- Other information
- Inactive ingredients
- Drug Facts
- Active ingredients
- Uses
-
Warnings
For external use only.
Do not use
• in the eyes • over large areas of the body • in large quantities • over raw surfaces or blistered areas • longer than 1 week unless directed by a doctor
- Directions
- Other information
- Inactive ingredients
- Drug Facts
- Active ingredient
- Uses
- Warnings
- Directions
- Other information
- Inactive ingredients
- Eye Wash (50814-010-01) Labeling:
- Antiseptic Towelettes (50814-011-01) Labeling:
- Alcohol Cleansing Pads (50814-012-01) Labeling:
- Antibiotic Application (50814-013-01) Labeling:
- Burn Treatment (50814-014-01) Labeling:
- Hand Sanitizer (50814-015-01) Labeling:
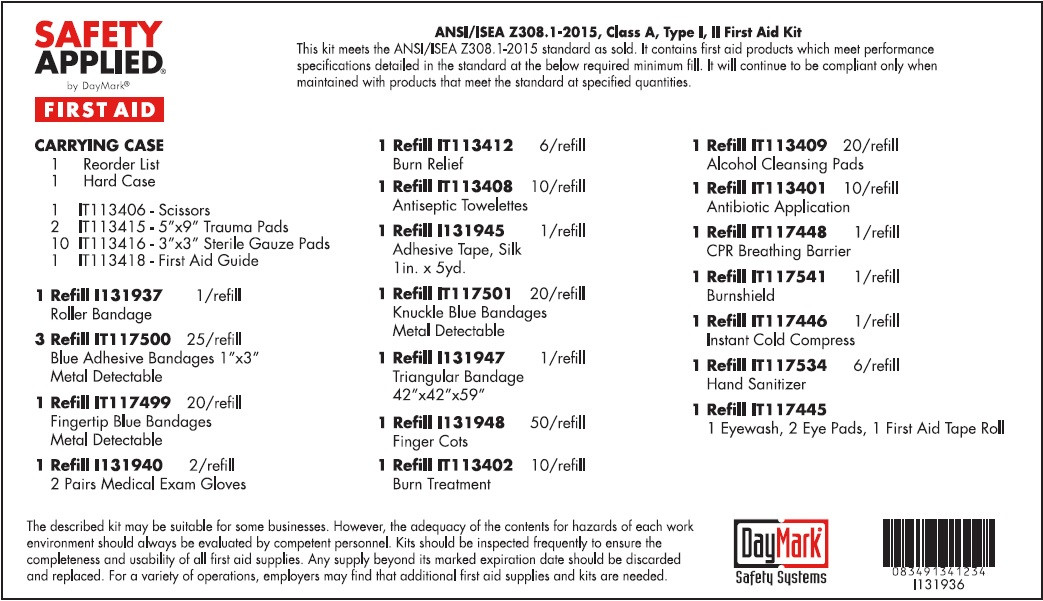
- Standard ANSI First Aid (50814-020-01) Labeling:
-
INGREDIENTS AND APPEARANCE
STANDARD ANSI FIRST AID
water, benzalkonium chloride, isopropyl alcohol, bacitracin zinc, neomycin sulfate, polymyxin b sulfate, lidocaine hydrochloride kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:50814-020 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-020-01 1 in 1 KIT 08/10/2016 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 TUBE 30 mL Part 2 10 PATCH 9 g Part 3 10 PATCH 9 g Part 4 6 PACKAGE 5 g Part 5 10 PACKAGE 9 g Part 6 6 PACKAGE 5.4 g Part 1 of 6 EYE WASH
water solutionProduct Information Item Code (Source) NDC:50814-010 Route of Administration OPHTHALMIC Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength WATER (UNII: 059QF0KO0R) (WATER - UNII:059QF0KO0R) WATER 991 mg in 1 mL Inactive Ingredients Ingredient Name Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) SODIUM CHLORIDE (UNII: 451W47IQ8X) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-010-01 1 in 1 BOX 1 30 mL in 1 TUBE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part349 08/10/2016 Part 2 of 6 ANTISEPTIC TOWELETTES
benzalkonium chloride clothProduct Information Item Code (Source) NDC:50814-011 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-011-01 1 in 1 BOX 1 10 in 1 BOX 1 0.9 g in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/10/2016 Part 3 of 6 ALCOHOL CLEANSING
isopropyl alcohol clothProduct Information Item Code (Source) NDC:50814-012 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 70 g in 100 g Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-012-01 1 in 1 BOX 1 10 in 1 BOX 1 0.9 g in 1 PATCH; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/10/2016 Part 4 of 6 ANTIBIOTIC APPLICATION
bacitracin zinc, neomycin sulfate, polymyxin b sulfate ointmentProduct Information Item Code (Source) NDC:50814-013 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BACITRACIN ZINC (UNII: 89Y4M234ES) (BACITRACIN - UNII:58H6RWO52I) BACITRACIN 400 [iU] in 1 g NEOMYCIN SULFATE (UNII: 057Y626693) (NEOMYCIN - UNII:I16QD7X297) NEOMYCIN 3.5 mg in 1 g POLYMYXIN B SULFATE (UNII: 19371312D4) (POLYMYXIN B - UNII:J2VZ07J96K) POLYMYXIN B 5000 [iU] in 1 g Inactive Ingredients Ingredient Name Strength MINERAL OIL (UNII: T5L8T28FGP) PETROLATUM (UNII: 4T6H12BN9U) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-013-01 1 in 1 BOX 1 10 in 1 BOX 1 0.9 g in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 08/10/2016 Part 5 of 6 BURN TREATMENT
benzalkonium chloride, lidocaine hydrochloride creamProduct Information Item Code (Source) NDC:50814-014 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength BENZALKONIUM CHLORIDE (UNII: F5UM2KM3W7) (BENZALKONIUM - UNII:7N6JUD5X6Y) BENZALKONIUM CHLORIDE 0.13 g in 100 g LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE 0.5 g in 100 g Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-014-01 1 in 1 BOX 1 10 in 1 BOX 1 0.9 g in 1 PACKAGE; Type 2: Prefilled Drug Delivery Device/System (syringe, patch, etc.) Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/10/2016 Part 6 of 6 HAND SANITIZER
alcohol gelProduct Information Item Code (Source) NDC:50814-015 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ALCOHOL (UNII: 3K9958V90M) (ALCOHOL - UNII:3K9958V90M) ALCOHOL 0.62 g in 1 g Inactive Ingredients Ingredient Name Strength CARBOXYPOLYMETHYLENE (UNII: 0A5MM307FC) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:50814-015-01 1 in 1 BOX 1 6 in 1 BOX 1 0.9 g in 1 PACKAGE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part333A 08/10/2016 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part333B 08/10/2016 Labeler - GFA Production (Xiamen) Co., Ltd. (421256261) Establishment Name Address ID/FEI Business Operations GFA Production (Xiamen) Co., Ltd. 421256261 manufacture(50814-020, 50814-010, 50814-011, 50814-012, 50814-013, 50814-014, 50814-015)