| NDC | 0363-9890-20 |
| Set ID | a016cf1c-a1b7-46a4-bda0-a5875e1be357 |
| Category | HUMAN OTC DRUG LABEL |
| Packager | Walgreens |
| Generic Name | |
| Product Class | alpha-1 Adrenergic Agonist, Antihistamine |
| Product Number | |
| Application Number | PART343 |
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 10 softgels in 24 hours, which is the maximum daily amount for this product
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy Alert
Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- If you have ever had an allergic reaction to this product or any of its ingredients
- In children under 12 years of age
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- cough with excessive phlegm(mucus)
- difficulty in urination due to enlargement of the prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Stop use and ask a doctor if
- pain, cough or nasal congestion gets worse or lasts more than 7 days
- new symptoms occur
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- cough comes back or occurs with rash or headache that lasts. These could be signs of a serious condition.
- nervousness, dizziness, or sleeplessness occurs.
-
Directions
- take only as recommended (see overdose warning)
- do not take more than recommended dose
- adults and children 12 years and over :take 2 softgels with water every 4 hours. Do not exceed 10 softgels in 24 hours or as directed by a doctor
- children under 12 years: do not use
- Other Information
- Inactive ingredients
- Questions or comments?
- SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Uses
-
Warnings
Liver warning
This product contains acetaminophen. Severe liver damage may occur if you take:
- more than 10 softgels in 24 hours, which is the maximum daily amount for this product
- with other drugs containing acetaminophen
- 3 or more alcoholic drinks every day while using this product
Allergy Alert
Acetaminophen may cause severe skin reactions. Symptoms may include:
- skin reddening
- blisters
- rash
If a skin reaction occurs, stop use and seek medical help right away.
Sore throat warning
If sore throat is severe, persists for more than 2 days, is accompanied or followed by fever, headache, rash, nausea, or vomiting, consult a doctor promptly.
Do not use to sedate children.
Do not use
- with any other drug containing acetaminophen (prescription or nonprescription). If you are not sure whether a drug contains acetaminophen, ask a doctor or pharmacist.
- if you are now taking a prescription monoamine oxidase inhibitor (MAOI) (certain drugs for depression, psychiatric, or emotional conditions, or Parkinson's disease), or for 2 weeks after stopping the MAOI drug. If you do not know if your prescription drug contains an MAOI, ask a doctor or pharmacist before taking this product.
- in children under 12 years of age
- If you have ever had an allergic reaction to this product or any of its ingredients
Ask a doctor before use if you have
- liver disease
- heart disease
- high blood pressure
- thyroid disease
- diabetes
- glaucoma
- cough with excessive phlegm (mucus)
- a breathing problem such as emphysema or chronic bronchitis
- difficulty in urination due to enlargement of the prostate gland
- persistent or chronic cough such as occurs with smoking, asthma, or emphysema
Ask a doctor or pharmacist before use if you are
- taking the blood thinning drug warfarin
- taking sedatives or tranquilizers
When using this product
- do not exceed recommended dosage
- may cause marked drowsiness
- avoid alcoholic drinks
- alcohol, sedatives, and tranquilizers may increase drowsiness
- be careful when driving a motor vehicle or operating machinery
- excitability may occur, especially in children
Stop use and ask a doctor if
- pain, cough, or nasal congestion gets worse or lasts more than 7 days
- fever gets worse or lasts more than 3 days
- redness or swelling is present
- new symptoms occur
- cough comes back or occurs with rash or headache that lasts. These could be signs of a serious condition.
- nervousness, dizziness, or sleeplessness occurs
- Directions
- Other information
- Inactive ingredients
-
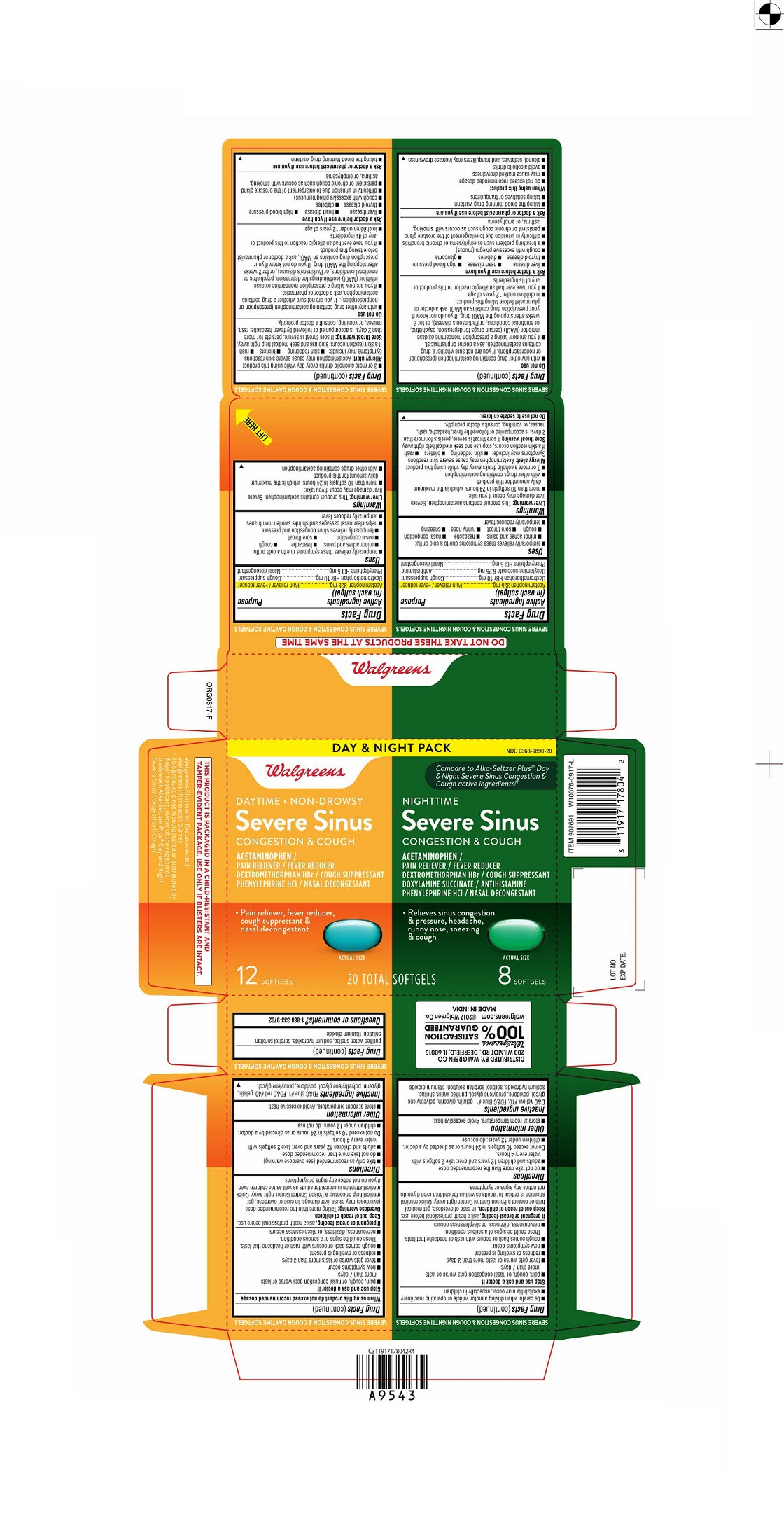
PRINCIPAL DISPLAY PANEL - Kit Carton
DAY & NIGHT PACK
NDC 0363-9890-20
Walgreens
Compare to Alka-Seltzer PLUS® Day and Night Severe Sinus Congestion & Cough active ingredients‡‡
DAYTIMENON-DROWSY
Severe Sinus
Congestion & CoughACETAMINOPHEN/
PAIN RELIEVER/FEVER REDUCER
DEXTROMETHORPHAN HBr/COUGH SUPPRESSANT
PHENYLEPHRINE HCL/NASAL CONGESTANT
- Pain reliever, fever reducer,
cough suppressant &
nasal decongestantActual Size
12 SOFTGELS
NIGHTTIME
Severe Sinus
Congestion & CoughACETAMINOPHEN/
PAIN RELIEVER/FEVER REDUCER
DEXTROMETHORPHAN HBr/COUGH SUPPRESSANT
DOXYLAMINE SUCCINATE/ANTIHISTAMINE
PHENYLEPHRINE HCL/NASAL DECONGESTANT
-
Relieves sinus congestion & pressure,
headache, runny
nose, sneezing &
cough
Actual Size
8 SOFTGELS
20 TOTAL SOFTGELS

-
INGREDIENTS AND APPEARANCE
SEVERE SINUS CONGESTION AND COUGH DAYTIME NON-DROWSY / NIGHTIME
acetaminophen, dextromethorphan hydrobromide, phenylephrine hydrochloride / acetaminophen, dextromethorphan hydrobromide, doxylamine succinate, phenylephrine hydrochloride kitProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:0363-9890 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:0363-9890-20 1 in 1 CARTON 12/15/2017 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 BLISTER PACK 12 Part 2 1 BLISTER PACK 8 Part 1 of 2 SEVERE SINUS CONGESTION AND COUGH DAYTIME NON-DROWSY
acetaminophen, dextromethorphan hydrobromide, phenylephrine hydrochloride capsule, liquid filledProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength FD&C BLUE NO. 1 (UNII: H3R47K3TBD) FD&C RED NO. 40 (UNII: WZB9127XOA) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POVIDONE K30 (UNII: U725QWY32X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SHELLAC (UNII: 46N107B71O) SODIUM HYDROXIDE (UNII: 55X04QC32I) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) Product Characteristics Color blue Score no score Shape OVAL (OBLONG) Size 20mm Flavor Imprint Code 314 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 12 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 08/15/2015 Part 2 of 2 SEVERE SINUS CONGESTION AND COUGH NIGHTIME
acetaminophen, dextromethorphan hydrobromide, doxylamine succinate, phenylephrine hydrochloride capsule, liquid filledProduct Information Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ACETAMINOPHEN (UNII: 362O9ITL9D) (ACETAMINOPHEN - UNII:362O9ITL9D) ACETAMINOPHEN 325 mg DEXTROMETHORPHAN HYDROBROMIDE (UNII: 9D2RTI9KYH) (DEXTROMETHORPHAN - UNII:7355X3ROTS) DEXTROMETHORPHAN HYDROBROMIDE 10 mg DOXYLAMINE SUCCINATE (UNII: V9BI9B5YI2) (DOXYLAMINE - UNII:95QB77JKPL) DOXYLAMINE SUCCINATE 6.25 mg PHENYLEPHRINE HYDROCHLORIDE (UNII: 04JA59TNSJ) (PHENYLEPHRINE - UNII:1WS297W6MV) PHENYLEPHRINE HYDROCHLORIDE 5 mg Inactive Ingredients Ingredient Name Strength POLYETHYLENE GLYCOL 400 (UNII: B697894SGQ) POVIDONE K30 (UNII: U725QWY32X) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WATER (UNII: 059QF0KO0R) SHELLAC (UNII: 46N107B71O) SODIUM HYDROXIDE (UNII: 55X04QC32I) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) D&C YELLOW NO. 10 (UNII: 35SW5USQ3G) FD&C BLUE NO. 1 (UNII: H3R47K3TBD) GELATIN (UNII: 2G86QN327L) GLYCERIN (UNII: PDC6A3C0OX) Product Characteristics Color green Score no score Shape OVAL (OBLONG) Size 20mm Flavor Imprint Code 116 Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 8 in 1 BLISTER PACK; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 08/15/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph not final part343 08/15/2015 Labeler - Walgreens (008965063) Registrant - Spirit Pharmaceuticals LLC (179621011) Establishment Name Address ID/FEI Business Operations MEDGEL PVT LTD 677385498 manufacture(0363-9890)